Login
Subscribeexperimental therapy

Canada Approves Experimental ALS Therapy
Andy Carstens | Jun 14, 2022 | 3 min read
The country’s provisional go-ahead could increase pressure already being exerted on the US Food and Drug Administration to approve the therapy.

Remdesivir Shows Promise in Largest of Several Clinical Trials
Catherine Offord | Apr 30, 2020 | 3 min read
Gilead’s experimental antiviral drug shortened the average time it took COVID-19 patients to recover in a NIAID-sponsored trial. There was weak evidence that it also helped reduce deaths.

Blood Pressure Meds Point the Way to Possible COVID-19 Treatment
Ashley Yeager | Apr 2, 2020 | 7 min read
There is little evidence that antihypertensive drugs worsen COVID-19, and scientists are instead exploring the idea that such medications—or their downstream effects—may actually alleviate symptoms.
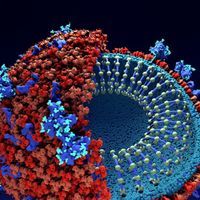
Remdesivir Works Against Coronaviruses in the Lab
Abby Olena, PhD | Mar 20, 2020 | 4 min read
The antiviral disables RNA replication machinery in MERS and SARS viruses. Can it beat back SARS-CoV-2?

Two Experimental Drugs Effective at Treating Ebola
Catherine Offord | Aug 13, 2019 | 2 min read
REGN-EB3 and mAb114 reduced mortality in a clinical trial carried out during the ongoing outbreak in Democratic Republic of Congo.

Experimental Ebola Treatments Approved for Use in Uganda
Catherine Offord | Jun 19, 2019 | 2 min read
The announcement comes as the country prepares for possible spread of the virus from Democratic Republic of Congo.

FDA Cracks Down on Purveyors of Stem Cell Treatments
Ashley P. Taylor | Dec 21, 2018 | 3 min read
The agency cites safety concerns with customers of Genetech who reported getting E. coli infections and sends letters to other stem cell companies reminding them to follow the rules.

Federal Trade Commission Takes Action Against Stem Cell Clinics
Ashley P. Taylor | Oct 19, 2018 | 2 min read
The FTC, in its first crack down on the industry, gets two California companies selling unproven treatments to stop their false advertising and pay a fine.

Ebola Update: More Experimental Treatments, Health Workers at Risk
Catherine Offord | Aug 23, 2018 | 2 min read
The number of cases in Democratic Republic of Congo’s latest outbreak of the virus has now surpassed 100.

More Vaccinations, Experimental Drugs as Potential Ebola Contacts Rise
Sukanya Charuchandra | May 30, 2018 | 2 min read
The World Health Organization predicts the number of cases in Democratic Republic of Congo will rise up to 300 by July.

Ebola Update: Funding, Vaccines, and More Deaths in DRC
Catherine Offord | May 23, 2018 | 2 min read
A total of 27 people have died since April, but new funds and the deployment of an experimental vaccine are expected to help contain the virus.

Biohacker Injects DIY Herpes Vaccine on Facebook Live
Shawna Williams | Feb 4, 2018 | 2 min read
The stunt, performed at a biohacking conference, was purportedly part of a two-subject trial of the genetically modified vaccine.

Funds To Speed Ebola Drug Development
Kerry Grens | Sep 4, 2014 | 1 min read
A $42 million US government contract awarded to an experimental Ebola medicine maker aims to accelerate the process of meeting demand for the therapeutic.

Experimental Ebola Drug Shows Promise
Tracy Vence | Sep 2, 2014 | 1 min read
ZMapp effectively rescued macaques from Ebola in a small trial, but it could be several months before supplies of the drug meet the growing human demand for it.

WHO OKs Experimental Ebola Drugs
Tracy Vence | Aug 13, 2014 | 2 min read
But biotech firms’ supplies are dwindling.

WHO Declares Ebola Emergency
Tracy Vence | Aug 8, 2014 | 1 min read
As the World Health Organization calls the ongoing Ebola outbreak in West Africa an international public health emergency, the US Food and Drug Administration green-lights the potential use of another experimental drug.

Bioethics of Experimental Ebola Treatments
Tracy Vence | Aug 7, 2014 | 3 min read
The administration of an experimental serum to two American healthcare workers treated for Ebola on US soil raises questions of safety, efficacy, access, and ethics.

Serum to Stop Ebola?
Kerry Grens | Aug 5, 2014 | 2 min read
An experimental monoclonal antibody therapy, tested only in animals, is given to two Americans infected with Ebola virus.
