This year, University of Paris-Saclay biologist Purificación López-García embarked with colleagues on a journey into life’s ancient past. The researchers traveled to the altiplanos of the northern Atacama Desert, high-altitude stretches of rocky soil and shrubbery in South America that are among the driest places in the world. Despite their inhospitable reputation, these plateaus may hold clues about the very origins of complex life. Amidst the dunes and barren mountains, there are pockets of life—warm, briny pools crusted over with colorful microbial mats of cyano-bacteria and archaea stacked atop one another like crepes. Long before Earth resembled its current state, López-García says, these microbial mats “were the forests of the past,” adding that scientists now use these clumps of microscopic life “as analogs of past ecosystems that certainly occurred at the time when eukaryotes first appear[ed].”
Each layer of these living mats is composed of different types of microbes that rely upon one another. At the surface, where light and oxygen are plentiful, photosynthesizing cyanobacteria dominate, while just below, heterotrophs that can persist in low-oxygen environments feed on their byproducts. Deeper down, the mats become dark and smelly, the result of the sulfate reducers and methanogens that populate these oxygen-bereft zones. Here, these partnerships become even more essential, with the castoffs of one group serving as fuel for another.
These close metabolic associations between organisms, a type of symbiosis known as syntrophy, may have prefaced the evolution of complex life by creating alliances that turned permanent over time, López-García says. In this way, individuals of different microbial species could have nested within one another to create a host with one or even several symbionts. This is exactly what scientists suspect happened to form a whole new type of cell, the eukaryote, which thrived and subsequently diversified into the macroscopic array of life we see today, including humans. So-called eukaryogenesis is not defined the same way by all researchers, but broadly, the term describes an evolutionary surge toward increasing cellular complexity between 1 and 2 billion years ago.
[Eukaryogenesis is] arguably one of the most important events in the history of life, after the origin of life itself.
—Daniel Mills, Ludwig-Maximilians-Universität München
During this time, some of the defining characteristics of modern eukaryotic cells—the nucleus, mitochondria, cytoskeleton, cell membrane, and chloroplasts, among others—made their debut. These occurred between the first and last common ancestors of all living eukaryotes, known by their acronyms, FECA and LECA, respectively. Most of the details of these evolutionary leaps, however, remain unsettled. Researchers do not uniformly agree on which branch of life eukaryotes sprang from, which microbial players might have contributed to the process, or on the order of specific evolutionary milestones along the way. But the recent identification of the Asgard archaea, thought to be the closest living relatives to modern eukaryotes, has enlivened discussions about eukaryogenesis.
Today, at the microbial mats in the Atacama Desert and other sites throughout the world, scientists are investigating what the earliest eukaryotic cells may have looked like, the partnerships they may have struck up with other organisms, and how their molecular machinery might have functioned and evolved. Already, the discovery of the Asgards has solidified certain aspects of eukaryogenesis while raising new questions about others. “I think this is the most exciting development in biology right now. So much is being discovered and so many predictions are being met,” says Daniel Mills, a geobiologist and postdoctoral researcher at Ludwig-Maximilians-Universität München who recently coauthored a paper suggesting that eukaryotes likely evolved in the absence of oxygen. Eukaryogenesis, he adds, is “arguably one of the most important events in the history of life, after the origin of life itself.”

Arrival of the Asgards
After receiving her PhD in 2013, evolutionary microbiologist Anja Spang was shopping around for a postdoc. For her dissertation, Spang had studied a group of archaea called the Thaumarchaeota (now Nitrososphaerota), and during that work, she’d picked up hints that the genomes of these and other archaea contained code for genes that produce what are known as eukaryotic signature proteins, or ESPs. These proteins should not have had recognizable counterparts in archaea, and yet, there they were. Wanting to understand just what was going on, Spang joined the lab of Thijs Ettema, an evolutionary microbiologist then at Uppsala University in Sweden, and set out in search of new data.
The team extracted genomes from sediments collected during a research cruise to a deep-sea vent site called Loki’s Castle located more than 2,300 meters below the surface of the Arctic Ocean, between Greenland and Norway. Ettema told The New York Times that the initial sample amounted to less than a teaspoonful of deep-sea muck. But almost immediately, software responsible for annotating and analyzing the genetic material began to return odd results—it flagged ESP homologs for actin, a distinctly eukaryotic protein that gives cells their shape, in a genome that was otherwise clearly archaeal. The microbes turned out to be members of a new group that Spang and the team named the Lokiarchaeota when they published their findings in Nature in 2015. In the years that followed, the team continued to flesh out this branch of the archaeal family tree, leading to the establishment of the Asgard superphylum, which in addition to Lokiarchaeota includes nods to other Norse gods, including the Thor-, Odin-, and Heimdallarchaeota.
Researchers have since identified other ESPs in these groups, including homologs of proteins involved in everything from ubiquitin signaling to gamete fusion. That ESPs are so common among Asgards suggests that these microbes represent the closest living prokaryotic relatives to modern eukaryotes and that modern eukaryotes may well have inherited aspects of their molecular machinery from archaea. Indeed, most scientists now argue that an ancient Asgard or another archaeon, and not a bacterium or proto-eukaryote as many previously assumed, likely served as the first host in the evolutionary process that ultimately resulted in a new type of cell.
In 2019, researchers successfully cultured an Asgard archaeon for the first time, allowing scientists to dive deeper into their biology. Using microscopy, Hiroyuki Imachi of the Japan Agency for Marine-Earth Science and Technology and colleagues found that the cultured species, for which they proposed the name Candidatus Prometheoarchaeum syntrophicum, is small and extremely slow-growing, dividing only every two to three weeks; some microbes can double in as little as a few minutes or hours. In addition, they found that Ca. P. syntrophicum lives in close association with another archaeon called Methanogenium. Ca. P. syntrophicum gets its energy by digesting amino acids and peptides for their nitrogen, and in turn, Methanogenium uses the hydrogen produced during that process to create its own fuel and at the same time reduce environmental hydrogen, which can induce cellular stress. This partnership confirms that Asgards engage in the type of relationships that researchers suspect gave rise to eukaryotes.
Hints of such a syntrophic relationship had been gleaned from other archaeal genomes, says Spang, who now oversees her own research group at the Royal Netherlands Institute for Sea Research, but Ca. P. syntrophicum provides tangible evidence. “I was really happy when I heard” of the preprint that first described the organism and its syntrophic lifestyle, she says. “[It] verified that at least the metabolic predictions for the Asgards were making sense with actual experimental work.”
The Path to ComplexityEukaryogenesis is broadly defined as the evolutionary path taken by increasingly complex lifeforms as they diverged from the simpler prokaryotes that dominated the early part of Earth’s biological history. The functional period of eukaryogenesis started just prior to the symbiosis between two prokaryotes and ended when the last common ancestor of modern eukaryotes arose. During this time, many of the most recognizable eukaryotic features appeared, including organelles such as mitochondria, nuclei, and chloroplasts, as well as cellular processes such as phagocytosis. The ordering of these events in time remains unclear.  © NICOLLE FULLER, SAYO STUDIO |
ORIGINAL HOST UNKNOWNWhile the identity of original host in the symbiotic partnership that birthed modern eukaryotic cells remains mysterious, some researchers say the evidence suggests it was an archaeon rather than a bacterium. Scientists call this host, which lived more than a billion years ago, the first eukaryotic common ancestor, or FECA. ORIGIN OF MITOCHONDRIAAt some point in the past, the prokaryote host formed a partnership with an alphaproteobacterium and permanently engulfed it, creating the mitochondrion. Researchers debate whether phagocytosis was needed to establish this relationship, but mitochondria did help power much of eukaryotes’ subsequent radiation. APPEARANCE OF UNIQUE FEATURESNumerous other features and processes associated with modern eukaryotic cells evolved during this time, including the nucleus and cytoskeleton. The order of their appearance is uncertain. BIRTH OF MODERN LIFEThe last eukaryotic ancestor (LECA) shared by all living eukaryotes today was already a complex cell by the time eukaryotes began to radiate. Over hundreds of millions of years, LECA gave rise to the complex organisms that exist today, including fungi, protists, plants, and animals. |
The many competing hypotheses of eukaryogenesis
These early observations precipitated a flood of new research, with hundreds of papers published as preprints on bioRxiv touching on Asgards and eukaryogenesis in the last several years. The most immediate effect of the discovery of Asgards was a shift in support from a three-domain tree of life that included eukaryotes, prokaryotes, and archaea to a two-domain model, often called the eocyte hypothesis, that lumps archaea and eukaryotes together. (See illustration.)
In the three-domain model, eukaryotes belong to a separate branch that shares a common ancestor with archaea. But phylogenetic analyses suggest that complex cells emerged from within the archaea. This results in two primary domains—bacteria and archaea—with eukaryotes being nested within archaea. “People were already arguing for a two-domain system before the Asgards were discovered, but then once the Asgards were described, it gave even more evidence,” says Andrew Roger, a molecular biologist at Dalhousie University in Nova Scotia. He adds that the two-domain hypothesis also “supports that the host during eukaryogenesis was an archaeon” and not a type of proto-eukaryote that formed a distinct lineage.
People were already arguing for a two-domain system before the Asgards were discovered, but then once the Asgards were described, it gave even more evidence.
—Andrew Roger, Dalhousie University
Researchers who spoke to The Scientist say that many scientists have rallied behind the idea that the first eukaryotes evolved out of a syntrophy between an archaeal host and bacteria that somehow found their way inside to become the organelles, such as nuclei and mitochondria, that distinguish eukaryotes. The details of these relationships remain murky, but mitochondria provide the most tantalizing clues to their origin story. “There’s DNA in mitochondria that we can somewhat clearly connect or trace back to alphaproteobacteria,” says Laura Eme, an evolutionary microbiologist at France’s National Centre for Scientific Research (CNRS). “Even if we don’t know exactly which lineage, we have a smoking gun.”
There are contrasting hypotheses as to how the alphaproteobacterium would have gotten inside an archaeal host, however. In the eukaryogenesis version of the chicken-and-egg conundrum, scientists go back and forth on whether mitochondria would have been necessary to power the energetically expensive process of phagocytosis, or whether phagocytosis would have had to arise first as the means of ingesting the symbiotic partner. An oscillation between “mito-early” and “mito-late” hypotheses appears frequently in the literature, but intriguingly, there were no known examples of phagocytosis in prokaryotes until very recently, when researchers identified a phagocytosis-like process of engulfment in a bacterium. “[M]any people were saying it is impossible to have the ancestor of mitochondria incorporated in any cell because phagocytosis is not known in the prokaryotic world,” says Eme. “Well, now we know that phagocytosis exists in bacteria, at least.”
Moreover, initial observations of the Asgards point to other mechanisms of engulfment. When scientists first cultured Ca. P. syntrophicum, they immediately noticed a series of thin projections coming off of the microbes—extensions of their membrane system called blebs. This observation suggested that these blebs might be able to surround an external entity—perhaps with the help of those actin homologs—and fuse together, trapping the foreign body inside. The phagocytosis conundrum “is much less of a problem now,” Eme tells The Scientist.

When it comes to the nucleus, what López-García calls “the typical diagnostic eukaryotic feature,” the picture is much less clear. Hypotheses of its origin run the gamut from a bacterial endosymbiont within an amoeboid host to the remnants of a giant virus. (See “From Three Domains to Two” below.) In the 1990s, López-García proposed the Syntrophy hypothesis for the origin of eukaryotes, which posited a three-party metabolic symbiosis between two bacteria and an archaeon. She maintains that this hypothesis is the only one that explains not only the origins of the nucleus, but also the so-called lipid divide, another unsettled aspect of eukaryogenesis in which the lipids that make up the cell membranes of eukaryotes are more similar to those in bacteria than to those in archaea.
A couple of years ago, López-García and her Paris-Saclay colleague David Moreira, also affiliated with the CNRS, updated the hypothesis to reflect the discovery of Asgards, but rather than place an archaeon as the original host, they propose than an archaeon—specifically a hydrogen-producing, Asgard-like archaeon—was the original nucleus. The host, they suggest, was likely a deltaproteobacterium, and the ancestor of mitochondria an alphaproteobacterium. This idea is supported, they say, by the fact that most genes in modern eukaryotes are actually bacterial, and not archaeal, in origin, and that eukaryotic membranes are made up of phospholipids that more closely resemble bacterial ones. “Our model is one potential model—it may be wrong, [or it] may be right—but the others don’t explain these discrepancies,” López-García says. “And at some point, I think they should.”
Michelle Leger, a postdoctoral researcher and evolutionary microbiologist at the Institute of Evolutionary Biology in Barcelona, is currently scouring the genomes of extant archaeal species to support or refute the many hypotheses floating around. With respect to the Syntrophy hypothesis, for example, “if I were to imagine that there was the deltaproteobacteria in that relationship as well, I would expect a similarly clear [genomic] signal” to that of the alphaproteobacteria in the mitochondrial genome, Leger tells The Scientist. She hasn’t found such a signal yet, but she says she thinks the evidence does support an archaeal origin for the nucleus. Although archaeal genes make up a small fraction of the nuclear genome, the genes that play roles in highly conserved processes within the nucleus itself, such as DNA replication and transcription, are largely archaeal. So “it makes sense” that the nucleus developed from an archaeon, Leger says. “But it’s not very clear what other partners might have been involved.”
New approaches to understanding eukaryogenesis
Even as the number of sequenced archaeal and bacterial genomes continues to increase, offering new clues about the relationship between these microbes and the rise of early eukaryotic cells, many researchers tell The Scientist it’s entirely possible that some questions will never be fully answered. Too much time has passed since eukaryotes first appeared on the evolutionary scene, and too much DNA has been scrambled between too many groups, for scientists to piece everything together. But that hasn’t stopped them from trying.
Eme tells The Scientist that the “next big frontier” will be functional studies in modern eukaryotes to yield clues about how individual genes and proteins may have behaved in their early ancestors. While there was only a single Asgard genome a few years ago, today there are hundreds, and researchers are mining them for details. “Now we have a clear idea of which genes in eukaryotes have been inherited from Asgard archaea, and there’s a lot of novelty here,” Eme says. “But what we don’t know, and that’s really important, is what these genes did or are doing in Asgard currently.”
From Three Domains to TwoThe question of where exactly eukaryotes branch on the tree of life has been debated by scientists for decades. But the discovery of the Asgard archaea—the closest prokaryotic relatives to modern eukaryotes—has shifted most researchers away from a three-domain tree in which eukaryotes are a distinct lineage and toward a two-domain tree, in which eukaryotes emerged from within the archaea as a secondary domain. 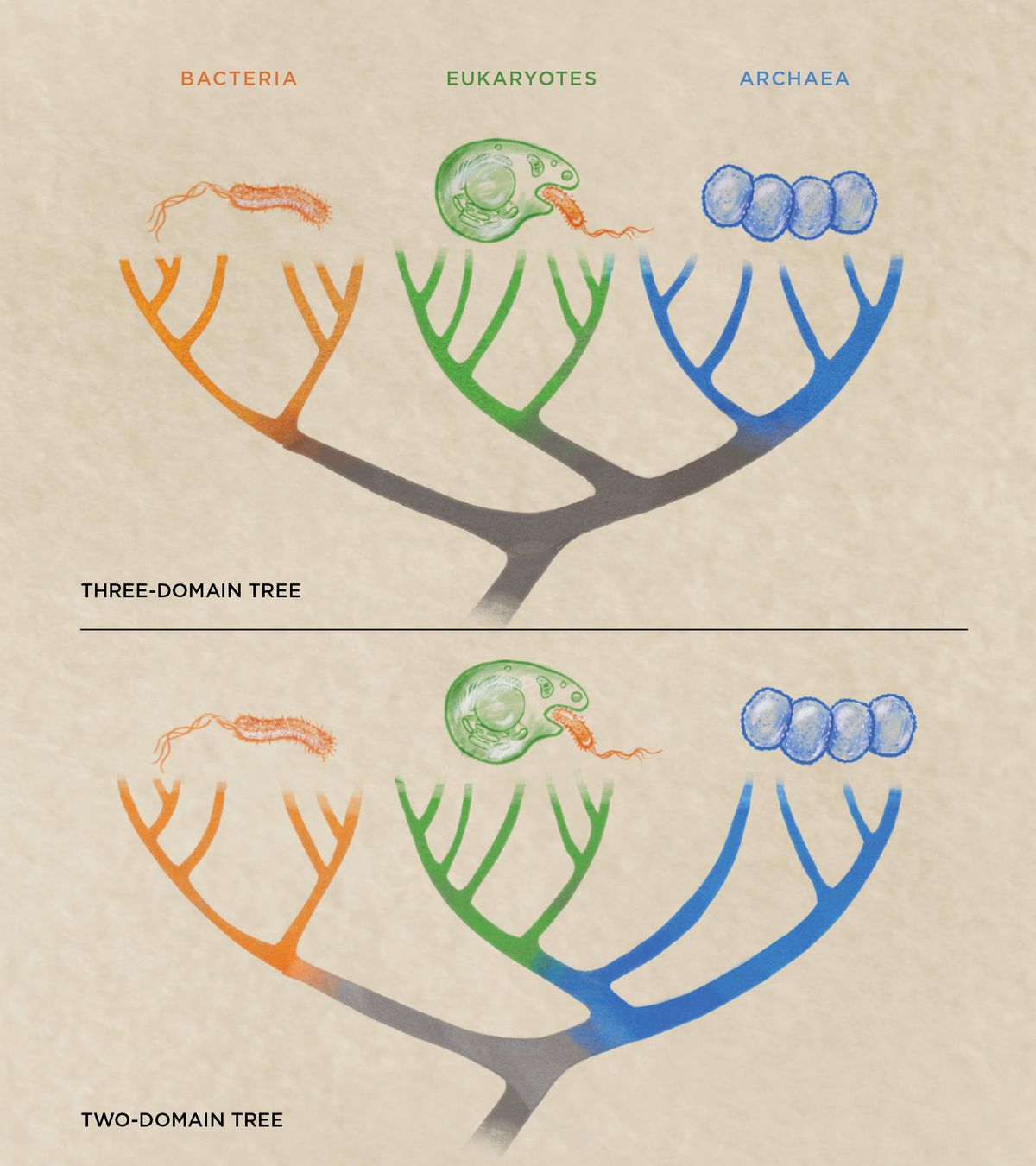 © NICOLLE FULLER, SAYO STUDIO |
In 2020, researchers synthesized suspected homologs of eukaryotic actin proteins encoded in Asgard genomes. Injected into rabbit cells, these proteins bound to eukaryotic actins and performed similar functions, including aiding the flow of calcium across cell membranes. The findings suggest that a calcium-controlled actin cytoskeleton likely existed in Asgards prior to the emergence of eukaryotes. In another study, researchers attempted to resolve the lipid debate by expressing archaeal phospholipids in E. coli, and found that the bacteria were able to successfully incorporate as much as 30 percent of the archaeal lipids into their cell membranes. The study doesn’t fully reconcile whether eukaryotes would have been able to transition their membranes from bacterial to archaeal lipids—López-García notes that bacteria with membranes composed of more than 30 percent archaeal lipids begin to die—“but it does lay the groundwork for future research,” Eme says.
Additional clues could come from the study of microfossils, microscopic impressions of early cells embedded in rock, says University of California, Santa Barbara, paleontologist Susannah Porter. When metagenomic sequencing came to the fore, it seemed as though fossils fell out of favor, she says, but many phylogenetic trees rely on a methodology called a molecular clock that uses fossils to anchor analyses in time. In addition, the fossils themselves can be useful, allowing scientists to determine when certain external features first appeared, adds Porter, who is currently interrogating such specimens to order certain events of early eukaryote evolution. “We do have a fossil record back 2 billion to 1 billion years, but I don’t think it’s been taken advantage of or leveraged to its full extent,” she says. “Maybe we could actually use these characteristics of the fossil record to be able to piece together eukaryogenesis.”
Meanwhile, other researchers are devising alternate methods for timing the events of eukaryogenesis to complement that fossil evidence. For example, Berend Snel, a computational biologist at Utrecht University in the Netherlands, recently used gene duplications to correlate the lengths of branches on phylogenetic trees with time—the assumption being that the number of duplication events increases with time. That assumption was challenged by some, and even Snel admits that “it may not be perfect,” but breaking the story of eukaryogenesis into more manageable chunks may help resolve many of these unanswered questions, he says. “What I’m arguing for is that it’s a lot of little, small stories, but if people would integrate these small stories in the right way, there should be a tapestry that ultimately weaves a real story.”
Leger agrees that our understanding of eukaryogenesis is likely to advance with baby steps. “Part of the nature of these deep evolutionary questions is that we will never know, we will never have a clear proof of some of the hypotheses that we’re trying to develop,” she says. “But we can keep refining our ideas.”
Viruses of the AsgardWhile much about the origin of the nucleus is speculative, one hypothesis suggests that the nucleus of modern eukaryotes may have resulted from a partnership between a prokaryotic host and a virus. This idea was first suggested in a pair of papers published back-to-back in 2001 after two researchers independently arrived at the same conclusion, and both groups recently published updates to their viral origin hypotheses following the field-rocking discovery of the Asgard archaea. At the turn of the 21st century, Masaharu Takemura, then a molecular biologist at the Nagoya University School of Medicine in Japan, noticed that one group of viruses, the poxviruses, had DNA polymerases that were extremely similar to those found in eukaryotes, and that poxviruses replicate inside their hosts by creating self-contained compartments. Meanwhile, Philip Bell, the head of research for the biotechnology company MicroBioGen, was similarly puzzled by the differences between eukaryotes and the bacteria that led to organelles such as mitochondria. Eukaryotic chromosomes are linear, for example, while bacterial ones are circular. Many features of the nucleus just didn’t support a bacterial origin. Since that time, researchers have identified the so-called giant viruses, first described in 2003. These viruses are much larger than most, with fittingly massive genomes, and they’ve since been found to harbor genes associated with various metabolic processes. Now, Takemura, Bell, and others say that a giant virus could have been the original nucleus. Giant viruses replicate within complex compartments that look very similar to modern nuclei—they’re large and include both inner and outer membranes—and also carry versions of genes that produce proteins involved in essential host cell processes. The idea that the nucleus could have been a virus has been a tough sell, however. According to Purificación López-García, a biologist at the University of Paris-Saclay, “there is no structural evidence” to support it. Michelle Leger, an evolutionary microbiologist at the Institute of Evolutionary Biology in Barcelona, agrees that the hypothesis is not supported by existing data, which she argues more clearly point to an archaeon as the organism that became the eukaryotic nucleus. But Valerie De Anda, a microbiologist at the University of Texas at Austin Marine Science Institute who studies early prokaryotic metabolism, isn’t dissuaded by the current lack of evidence from the idea that a virus may well be the source of the eukaryotic nucleus. She and her colleagues are currently looking for mRNA-capping genes involved in transcription and translation that were suggested by Bell to have been derived from a long-ago “first eukaryotic nuclear ancestor.” “People don’t take seriously great ideas right at the beginning . . . and then it turns out to be true,” De Anda says. |
Correction (October 18): This article has been updated to reflect that Valerie De Anda studies early prokaryotic metabolism, not early eukaryote metabolism, and to specify that eukaryotic signature proteins have been linked to gamete fusion, not meiosis. The Scientist regrets these errors.

