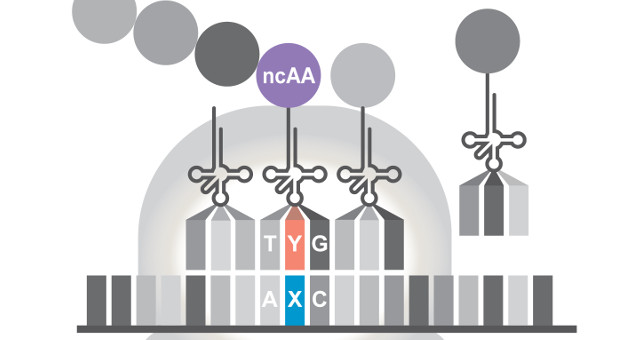
 ADAPTED FROM AN IMAGE BY DENNIS SUN, MEZARQUE DESIGNA synthetic base pair, first reported in 2014, can now not only replicate inside living cells, but also encode and produce proteins containing atypical amino acids, according to a report in Nature today (November 29). This proof-of-principle advance now sets the stage for biochemists to generate proteins with entirely novel forms and functions to those that can be created by natural organisms, say the authors.
ADAPTED FROM AN IMAGE BY DENNIS SUN, MEZARQUE DESIGNA synthetic base pair, first reported in 2014, can now not only replicate inside living cells, but also encode and produce proteins containing atypical amino acids, according to a report in Nature today (November 29). This proof-of-principle advance now sets the stage for biochemists to generate proteins with entirely novel forms and functions to those that can be created by natural organisms, say the authors.
“What a beautiful paper,” says chemical and biological engineer Michael Jewett of Northwestern University who was not involved in the study. “What’s so special about the work is that the authors have captured the entire information flow of the central dogma—information storage, retrieval, and, ultimately, translation into a functional output—using this expanded genetic alphabet.”
In all forms of life on earth, genetic information is composed of a four-letter alphabet—the nucleotides G, C, A, and T, which form the base pairs G-C and A-T. But three years ago, chemistry professor Floyd Romesberg of the Scripps Research Institute in California and colleagues extended this alphabet, reporting the creation of additional artificial nucleotides, X and Y, that could pair up within DNA and take part in replication within ...





















