Cold sores—painful blisters that pop up on lips seemingly out of nowhere—bother more than 60 percent of people under 50 years old. After the herpes simplex virus type 1 (HSV-1) virus, which causes cold sores, initially infects human cells in its lytic cycle, it can lay dormant in the body for many years. In these latent cycles, HSV-1 seeks refuge in nerve cells, which allows the virus to establish lifelong infections and makes it difficult to target using antivirals.1

Esther González-Almela is a virologist at the National Center of Biotechnology in Spain who studies the molecular mechanisms of viral infections. At the time of the study, she worked at the Guangdong Academy of Medical Sciences.
Esther González-Almela
“To completely cure HSV-1, the idea is to prevent the virus from going into latency, because then, it’s protected by our own bodies,” said Esther González-Almela, a virologist at the Guangdong Academy of Medical Sciences. “If we want to target this virus, it's better to do it during lytic cycle.”
In a recent study, González-Almela and her colleagues discovered a potential way to stop HSV-1 before it has a chance to hide in the body.2 They found that during HSV-1's lytic cycle, the virus takes over host cell’s enzymes, RNA polymerase II (RNAP II) and topoisomerase 1 (TOP1), for its own gene transcription and replication. When the researchers inhibited TOP1, these processes halted, suggesting that targeting this enzyme could be a potential therapeutic strategy. They published the results in Nature Communications.
“It’s a very fascinating and important study,” said Alex Evilevitch, a biophysicist at Lund University who was not involved in the research. “There’s a strong, unmet need for HSV-1 treatment, and now, there’s clearly an interesting new prospect.”
Researchers knew that when HSV-1 initially infects cells, it hijacks RNAP II to transcribe its genome.3 They also had evidence that the virus dramatically alters the host’s chromatin structure, compacting and marginalizing the host genome to the nuclear periphery.4-7
“But nobody knew how it was happening, or what was driving this compaction,” González-Almela said.
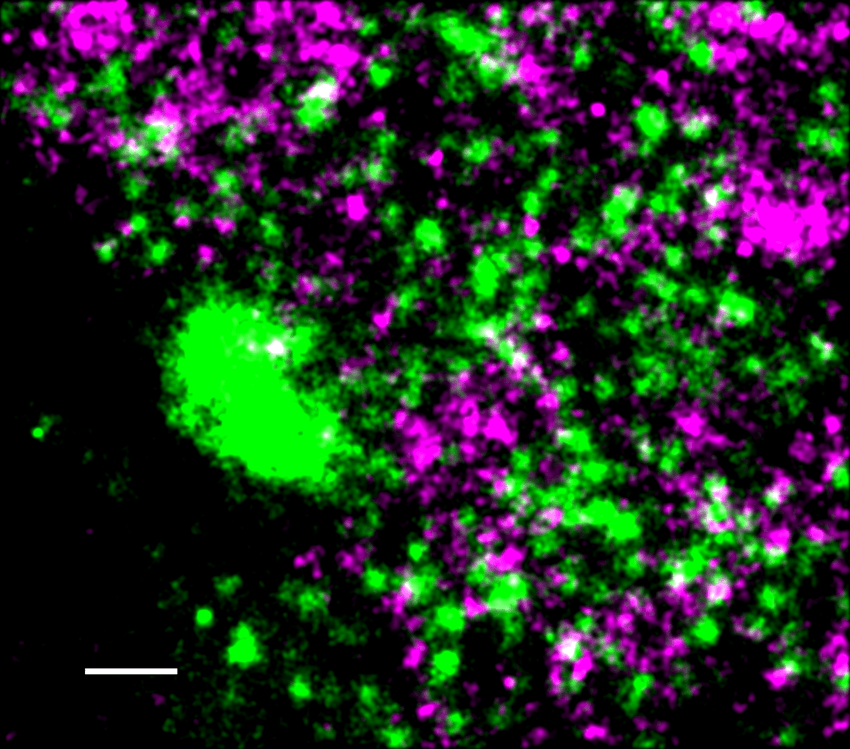
Active RNAP II (green) closely associates with viral DNA (magenta) in the VRC, as this super-resolution microscopy image demonstrates. Scale bar represents 500nm.
Esther González-Almela & Alvaro Castells-Garcia
To find out, González-Almela and her colleagues used super-resolution microscopy to distinguish between viral replication compartments (VRCs), the regions where viral gene transcription occurs, and the rest of the nucleus, which they called the host’s compartment (HC). By combining super-resolution imaging with chromatin analysis, the researchers demonstrated that viral DNA physically interacted with host DNA at transcriptionally active regions of the host genome.
Evilevitch said, “The most exciting thing for me was to see [viral-host] interactions in very specific [host genomic] regions so early in the infection process.”
The researchers showed that within three hours of HSV-1 infection, the virus completely sequestered the host’s RNAP II to VRCs. When the researchers infected cells with mutant HSV-1 that could not hijack RNAP II, genome compaction did not occur, indicating that RNAP II hijacking drove this phenomenon.
To see if HSV-1 hijacked other components of the host’s molecular machinery, the researchers performed differential gene expression analysis, where they found that TOP1, along with RNAP II, was increasingly upregulated as the infection progressed.

Alvaro Castells-Garcia is a molecular biologist at the Madrid Institute for Advanced Studies in Nanoscience. At the time of the study, he worked at the Guangdong Academy of Medical Sciences. He and González-Almela contributed equally to the study.
Alvaro Castells-Garcia
“Our discovery of TOP1 was a bit serendipitous,” González-Almela said.
Using the same super-resolution microscopy techniques, the researchers showed that HSV-1 hijacked TOP1 from host DNA and sequestered it to VRCs, much like it did RNAP II. They also found that when they treated cells with β-lapachone, a TOP1 inhibitor, both the transcription and replication of viral genes halted.
González-Almela hopes that TOP1 could serve as an antiviral drug target, but David Knipe, a virologist at Harvard Medical School who was not involved in the study, cautioned, “Inhibiting a host cell enzyme is potentially very toxic.”
In the future, González-Almela and her colleagues want to know what triggers HSV-1 to wake up from long periods of latency and if the molecular mechanisms that are crucial for its initial infection are involved.
- Knipe DM, Cliffe A. Chromatin control of herpes simplex virus lytic and latent infection. Nat Rev Microbiol. 2008;6(3):211-221.
- González-Almela E, Castells-Garcia A, et al. Herpes simplex virus type 1 reshapes host chromatin architecture via transcription machinery hijacking. Nat Commun. 2025;16(1):5313.
- Abrisch RG, Eidem TM, Yakovchuk P, Kugel JF, Goodrich JA. Infection by herpes simplex virus 1 causes near-complete loss of RNA Polymerase II occupancy on the host cell genome . J Virol. 2015;90(5):2503-2513.
- Simpson-Holley M, et al. Identification and functional evaluation of cellular and viral factors involved in the alteration of nuclear architecture during herpes simplex virus 1 infection. J Virol. 2005;79(20):12840-12851.
- Monier K, et al. Annexation of the interchromosomal space during viral infection. Nat Cell Biol. 2000;2(9):661-665.
- Aho V, et al. Infection-induced chromatin modifications facilitate translocation of herpes simplex virus capsids to the inner nuclear membrane. PLoS Pathog. 2021;17(12):e1010132.
- Evilevitch A, Hohlbauch SV. Intranuclear HSV-1 DNA ejection induces major mechanical transformations suggesting mechanoprotection of nucleus integrity. Proc Natl Acad Sci U S A. 2022;119(9):e2114121119.













