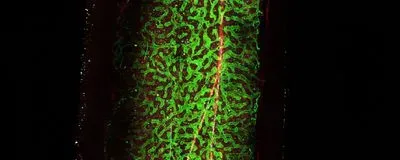
ABOVE: Deep imaging of bone marrow in a mouse femur showing that osteolectin-expressing cells (red) are around arterioles (white) but not sinusoids (green), a different type of blood vessel in the bone marrow.
CHILDREN’S MEDICAL CENTER RESEARCH INSTITUTE AT UT SOUTHWESTERN
Mechanical forces from running and walking that are transmitted along blood vessels in marrow induce the growth of new bone and immune cells in mice, scientists reported in Nature on February 24. The study is the first to demonstrate that mechanical forces can influence cellular growth and differentiation in the bone marrow, according to the authors, and provides a possible new explanation for how exercise strengthens bones and the immune system.
It’s well known that aging weakens bones and running can help strengthen them. “The way that’s understood to work is the mechanical forces are thought to act on the bone itself. And the soft bone marrow inside your bones ...