It began with little pieces of embryos scooting around in a dish. In 1998, these unassuming cells caught the attention of Michael Levin, then a postdoctoral researcher studying cell biology at Harvard University. He recalled simply recording a video before tucking the memory away. Nearly two decades later, Levin, now a developmental and synthetic biologist at Tufts University, experienced a sense of déjà vu. He observed that as a student transplanted tissues from one embryo to another, some loose cells swam free in the dish.
Levin had a keen interest in the collective intelligence of cells, tissues, organs, and artificial constructs within regenerative medicine, and he wondered if he could explore the plasticity and harness the untapped capabilities of these swirling embryonic stem cells. “At that point, I started thinking that this is probably an amazing biorobotics platform,” recalled Levin. He rushed to describe this idea to Douglas Blackiston, a developmental and synthetic biologist at Tufts University who worked alongside Levin.
At the time, Blackiston was conducting plasticity research to restore vision in blind African clawed frog tadpoles, Xenopus laevis, a model organism used to understand development. Blackiston transplanted the eyes to unusual places, such as the back of the head or even the tail, to test the integration of transplanted sensory organs.1 The eye axons extended to either the gut or spinal cord. In a display of dynamic plasticity, transplanted eyes on the tail that extended an optic nerve into the spinal cord restored the tadpoles’ vision.2
Levin and Blackiston decided to explore this remarkable ability to adapt to changes in function and connectivity, a key feature for regenerative medicine applications. By leveraging nature’s proficiency in building and rebuilding, they wanted to understand the limits of cell and tissue plasticity outside of their natural contexts to perform new functions.
It’s more like craftsmanship than it is science at times because you’re doing very fine manipulations.
—Douglas Blackiston, Tufts University
In a similar vein, Josh Bongard, an evolutionary roboticist at the University of Vermont and Levin’s longtime colleague, pondered how robots could evolve like animals. He wanted to apply biological evolution to a machine by tinkering with the brains and bodies of robots and explored this idea with Sam Kriegman, then a graduate student in Bongard’s group and now an assistant professor at Northwestern University. Kriegman used evolutionary algorithms and artificial intelligence (AI) to simulate biological evolution in a virtual creature before teaming up with engineers to construct a physical version.
Levin’s biology and Bongard’s computational work intersected for a program called Lifelong Learning Machines (L2M). With this project, the researchers aimed to understand how biological systems adapt to their environments and integrate these living algorithms into robotics.
Together, the team dovetailed developmental biology using different biological tissues as the building blocks and AI programs to generate synthetic lifeforms as the blueprints for biological robots (biobots), also known as xenobots, from frog cells. Over time, scientists incorporated cilia into these biodegradable xenobots, giving them the ability to swim. While researchers currently use xenobots as a tool to understand how cells cooperate to build complex systems, these could also have potential environmental applications: They could sense pollutants and gather or break down harmful materials like microplastics.
The Birth of Xenobots
At the beginning of this project, the team planned to build lifelong learning machines from AI systems, which was a challenging enterprise. Kriegman used evolutionary algorithms to design and evolve synthetic lifeforms in simulation, but the major stumbling block lay in translating these designs to the physical world. During weekly virtual meetings between the biologists and roboticists, Bongard recalled explaining to the biologist group what was easy and hard for roboticists to do; the conventional materials used to construct robots weren’t working.

Josh Bongard, Michael Levin, Douglas Blackiston, and Sam Kriegman (left to right) teamed up to build synthetic organisms called xenobots with an unlikely building material: frog stem cells.
DOUGLAS BLACKISTON AND SAM KRIEGMAN, LICENSED UNDER CC BY 4.0.
“It’s really difficult to realize [this idea] in hardware; no one’s figured out how to create a robot that crawls out of a 3D printer,” explained Kriegman. “We tried to build robots out of rubber, 3D printers, and electronics, but there’s always this problem. It’s very difficult.”
As Kriegman presented a video of little blob-like robots running around in a virtual environment, he described this challenge to the team. Within the computer simulations, these robots could be manipulated like a video game; it was easy to simulate physics principles like friction or modulate the virtual environment. However, the roboticists didn’t think that they could translate these theoretical designs and simulations into the real world with the existing tools.
Blackiston rose to the challenge. He conceptualized a virtual robot built out of a different material: cells. “In developmental biology and stem cell biology, this isn’t a super difficult trick because the technology exists, but no one’s thought about doing this,” said Blackiston.
Blackiston got to work in the laboratory using extra cells from his X. laevis project. Through delicate micromanipulations of stem cells in the microscope room, he crafted a replica of Kriegman’s virtual creature. About a week after Kriegman shared his simulations, Blackiston revealed his creation, affectionately dubbed the Bongard-bot, in a Slack thread. These robots would eventually come to be known as xenobots in popular culture.
“When Sam and I were looking at this image, we weren’t sure what we were looking at. It looked like Sam’s virtual robot that he had shown the week before, but it was clearly made of cells,” said Bongard. Although it was a rough approximation, floating in freshwater at only a few millimeters wide, it matched the virtual design.
While this creation emerged as an unexpected tangent to the initial L2M goal, it quickly became clear that this approach could breathe life into their simulations. Levin and Bongard encouraged Blackiston and Kriegman to explore this whole new space, moving between running thousands of simulations and sculpting the best designs out of cells. “From there on, it was off to the races,” recalled Kriegman.
A Xenobot Takes its First Steps
Since the initial xenobot remained static, the team wanted to see if they could make the newer version move. Kriegman initiated the iterative design of synthetic living machines by using AI to create virtual creatures; these innocuous blobs shuffled along the floor of a virtual world before gradually developing proto-legs or -arms. Then he and Blackiston selected the most viable designs to construct out of frog cells.

Xenobots are AI-designed organisms (red) crafted from frog stem cells (green).
DOUGLAS BLACKISTON AND SAM KRIEGMAN, LICENSED UNDER CC BY 4.0.
In his initial simulations for locomotion, Kriegman based the iterations on frog skin and heart cells given their propensities to aggregate and contract, respectively.3 With heart cells, they hoped to leverage motor movements from the heart muscle, like a piston, that would coordinate a form of locomotion.
Kriegman needed the computer to determine the optimal position and shape for these cellular motors in the xenobots. However, there was no guarantee that the evolved simulation would be feasible in the real world. With limited information, Kriegman sought the expertise of heart researchers to gain some insights into heart cell synchronization and to learn how unconventional shapes may influence cellular function. “We know how these cells work in the heart shape, but what would happen with these cells in the context of xenobots?” he wondered. “It’s difficult to predict ahead of time, so the AI or evolutionary algorithm must find designs that work regardless of how the motors are moving. It’s making reliable machines with unreliable parts.”
The team had to get creative during this process. “Based on the AI, Doug would build it, and then they would modify the AI and build the next iteration,” recalled Levin. “Going back and forth, it was amazing because every week there was something new to look at.”
It’s a great reminder that when it comes to robotics and AI, humans tend to overthink things. It’s better to let evolution, either biological or artificial, figure out how much complexity is needed for the task at hand.
—Josh Bongard, University of Vermont
As Kriegman generated numerous iterations of xenobots, Blackiston used the 3D image as the working blueprint in his microscope room. Blackiston gathered his ingredients using different biological tissues harvested from blastula stage X. laevis embryos. Then, as if building a sandwich, he arranged the different cell layers one at a time into a cube of tissue.
When the tissue healed together, it formed a sphere. Then Blackiston sculpted the tissue using a microsurgical tool with a wire smaller than a human hair to achieve the desired shape. Each cell type differed by color, and he rotated between filters to maintain the correct orientation. “It’s more like craftsmanship than it is science at times because you’re doing very fine manipulations,” remarked Blackiston. The final product resembled a speck of pepper moving in a petri dish. These biodegradable xenobots lived for about a week, sustaining themselves on their own food source (a yolk of lipid and protein deposits) before they degraded and ceased functioning.
One of the designs featured heart muscle cells on the bottom and skin cells on top with two stumpy legs on one side. As a result, it leaned over on its chest and could “walk” by moving forward in a straight line. However, when flipped onto its back, the simulated design became immobile due to the alteration in shape and tissue distribution. To verify whether the computer-generated in silico xenobot simulation matched what was created in the laboratory, Kriegman compared the trajectories of the physical xenobot and those of the virtual xenobot. To the team’s surprise, the two trajectories almost perfectly overlapped with one another. “It wasn’t just for one trajectory; there were lots of pairs,” recalled Bongard. “It confirmed that what happened in simulation matched what happened in reality.”
Xenobot Walked so Xenobot 2.0 Could Swim
The team next wanted to see if they could make a xenobot swim. To do this, the researchers employed another type of motor: cilia.4 Instead of layering different tissues, Blackiston used whole explants from developing frog embryos.
These explants, known as animal caps, have been used to study cell differentiation and tissue formation.5 The team repurposed the animal cap to create living machines with new specific functions. Once the X. laevis explants balled up into a spherical mass, they gained motility from cilia, which propelled them through their aqueous environments.
While their movements were less predictable compared to their walking counterparts, these ciliated xenobots could navigate. The xenobots swam through open fields, mazes, and even narrow capillaries. In environments with debris or silicone-coated beads, xenobots collectively swarmed together to push the debris into piles.
Not only did the xenobots demonstrate self-locomotion, but they could also be modified to record an experience. The team tested the biobots’ ability to sense their environments by microinjecting mRNA with a photoconvertible reporter that caused them to fluoresce green. Xenobots freely explored their surroundings, but if any xenobot passed through an area exposed to blue light, the reporter underwent a permanent conformational change, causing the xenobot to glow red. Otherwise, xenobots glowed green to indicate that they did not detect the blue light.
Xenobots also exhibited inherent robustness and could automatically self-repair after injury from surgical forceps. Every single xenobot could close a wound, resolve the injury, and reform into a spherical shape within minutes. “From a robotics perspective, cells are like a technology from a thousand years in the future that have just been plopped on our desks. They work when you stick them together. They survive; they’re self-powered; and they heal,” remarked Kriegman.
Xenobots Building Xenobots
As they watched the xenobots scoot and shuffle across the dish and push debris into piles, the researchers envisioned adding another feature. If the debris was replaced with other materials such as stem cells or even microplastics, the ability to collect materials could open up new areas for xenobot applications. They just needed a new design.
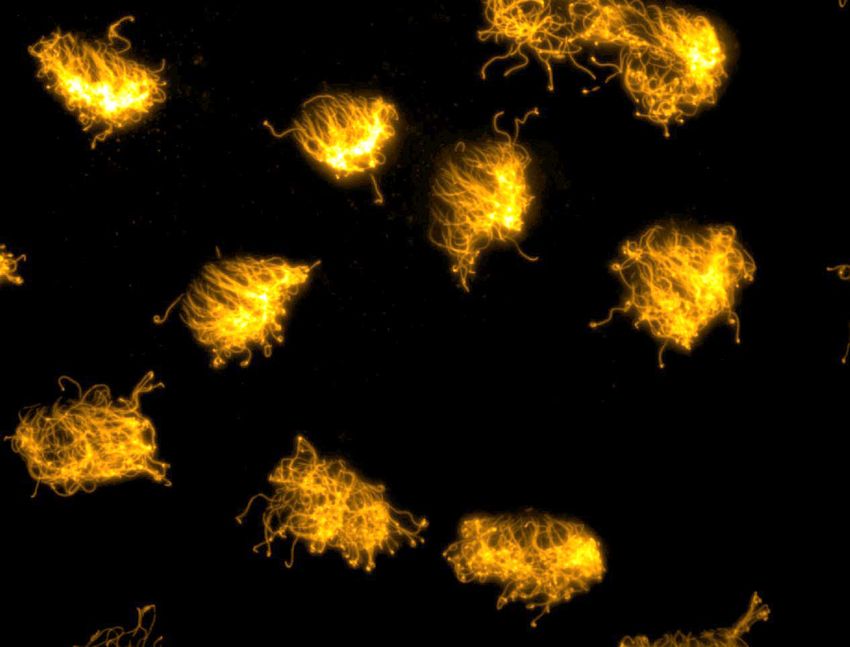
The presence of cilia, marked in orange fluorescence, enabled xenobots to swim in their environments.
DOUGLAS BLACKISTON AND SAM KRIEGMAN, LICENSED UNDER CC BY 4.0.
The initial spheroid shape wasn’t the best for this task. When Kriegman returned to the computer drawing board, he was surprised by the simplicity of the suggested design: a C-shape. “It’s a great reminder that when it comes to robotics and AI, humans tend to overthink things. It’s better to let evolution, either biological or artificial, figure out how much complexity is needed for the task at hand,” said Bongard.
This C-shape, reminiscent of Pac-Man or a snowplow, led to an unexpected discovery. C-shaped xenobots “reproduced” spontaneously in a manner dubbed as kinematic self-replication.6 When the team replaced debris with loose, white-colored stem cells, the xenobots immediately set to work collecting cells.
Over time, the piles of collected cells grew big enough to begin swimming themselves. These baby xenobots, although smaller than their makers, were created without evolution or genetic manipulation. Interestingly, this process occurred entirely within the dish. If there weren’t enough loose cells around, self-replication ceased; parent xenobots could only produce a round or two of self-replication before petering out.
The concept of kinematic self-replication was first proposed in the 1940s by a mathematician named John von Neumann.7 In this hypothetical model, a machine could assemble parts to create a duplicate of itself. However, true replication only occurs in nature, while machine replication is limited to generating computer viruses. “[With xenobots], this is a new way that people haven’t thought about where biological systems, namely cell clusters, can replicate,” said Kriegman. “Maybe this will help people think differently about replication.”
By leveraging existing techniques, the team built something that was not found in nature and reconfigured it to fulfill a new function. These xenobots have challenged conventional categories: Are they robots, living things, or machines? While the categorization of these synthetic living organisms may need to be redefined into a new box altogether, one thing remains certain: the team has only scratched the surface of xenobots’ capabilities.
“It’s a green technology in every sense of the word. What is the probability that it will never have an application? To me, I’m biased, but I think it’s close to zero. It’s going to find a use somewhere, but who knows how many uses and how long it will take?” said Bongard.
People thought this was a one-off froggy specific result, but this is a very profound thing. What’s the furthest from an embryonic frog? Well, that would be an adult human.
—Michael Levin, Tufts University
Potential avenues for these biodegradable machines primarily revolve around environmental applications, from serving as biosensors to detect pollutants to gathering materials like microplastics or even sequestering and breaking down harmful chemicals.
Evolving Beyond the Xenobot
“People thought this was a one-off froggy-specific result, but this is a very profound thing,” emphasized Levin. To demonstrate its translatability in a non-frog model, he wondered, “What’s the furthest from an embryonic frog? Well, that would be an adult human.”
He enlisted the help of Gizem Gumuskaya, a synthetic biologist with an architectural background in Levin’s group, to tackle this challenge of creating biological robots using human cells to create anthrobots.8 While Gumuskaya was not involved with the development of xenobots, she drew inspiration from their design. By using adult human tracheal cells, she found that adult cells still displayed morphologic plasticity.

The resulting anthrobots swam using cilia, but they unexpectedly also moved across a layer of damaged human neurons. To Gumuskaya’s surprise, the anthrobots aggregated and formed what she described as an ant bridge between the two damaged edges. While how they accomplished this remains unknown, the anthrobots aided in healing the neuronal tear, indicating that they may offer therapeutic potential.
There are several key differences between xenobots and anthrobots: species, cell source (embryonic or adult), and the anthrobots’ ability to self-assemble without manipulation. “When considering applications, as a rule of thumb, xenobots are better suited to the environment. They exhibit higher durability, require less maintenance, and can coexist within the environment,” said Gumuskaya.
Meanwhile, there is greater potential for the use of mammalian-derived biobots in biomedical applications. This could include localized drug delivery, deposition into the arteries to break up plaque buildup, or deploying anthrobots into tissue to act as biosensors. “[Anthrobots] are poised as a personalized agent with the same DNA but new functionality,” remarked Gumuskaya.
Gumuskaya hopes that this work in frogs and human cells inspires the scientific community to explore the new and unexpected functionalities of these bioconstructs. “There are a lot of big challenges in this world, but we’re developing new kinds of technologies and tools for the next generation. I hope that these bots become one tool in that toolkit,” remarked Bongard.
Key Takeaways
- Xenobots are living robots derived from the stem cells of the African frog Xenopus laevis. They are built by combining stem cells with other biological tissues and can perform some desired functions.
- By incorporating heart cells or cilia, scientists developed xenobots with the ability of locomotion. Building such biobots from mammalian cells could potentially have applications in drug delivery, breaking plaque buildup in arteries, or as biosensors.
- Scientists still debate whether xenobots are robots, living things, or machines. Many think that these synthetic living creatures may need to be redefined into a new category altogether.
- Xenobots are self-healing and can spontaneously “reproduce” to give rise to smaller, functional xenobots when provided with enough stem cells. Xenobots can find and combine loose stem cells into clusters, giving rise to baby xenobots.
- Blackiston DJ, Levin M. Ectopic eyes outside the head in Xenopus tadpoles provide sensory data for light-mediated learning. J Exp Biol. 2013;216(Pt 6):1031-40.
- Blackiston DJ, et al. Serotonergic stimulation induces nerve growth and promotes visual learning via posterior eye grafts in a vertebrate model of induced sensory plasticity. npj Regen Med. 2017;2:8.
- Kriegman S, et al. A scalable pipeline for designing reconfigurable organisms. Proc Natl Acad Sci USA. 2020;117(4):1853-1859.
- Blackiston D, et al. A cellular platform for the development of synthetic living machines. Sci Robot. 2021;6(52):eabf1571.
- Gallagher BC, et al. Autonomous differentiation of dorsal axial structures from an animal cap cleavage stage blastomere in Xenopus. Development. 1991;112(4):1103-1114.
- Kriegman S, et al. Kinematic self-replication in reconfigurable organisms. Proc Natl Acad Sci USA. 2021;118(49): e2112672118.
- von Neumann J, Burks AW. Theory of Self-Reproducing Automata. University of Illinois Press. 1966;418.
- Gumuskaya G, et al. Motile living biobots self-construct from adult human somatic progenitor seed cells. Adv Sci (Weinh). 2024;11(4):e2303575.















