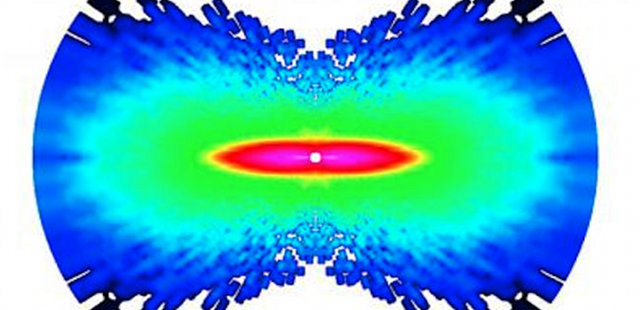
 A nitrogen molecule shifts during the time between laser pulses, one femtosecond. The atoms' movements are shown as a measure of increasing angular momentum, on a scale from dark blue to pink, with pink showing the region of greatest momentum. IMAGE COURTESY OF COSMIN BLAGA
A nitrogen molecule shifts during the time between laser pulses, one femtosecond. The atoms' movements are shown as a measure of increasing angular momentum, on a scale from dark blue to pink, with pink showing the region of greatest momentum. IMAGE COURTESY OF COSMIN BLAGA
THE DEVICE: Molecules are in constant motion as electrons swarm around inside atoms, slightly flexing the atomic bonds and shifting the molecule’s shape. Using standard tools found in physical chemistry labs, physicist Cosmin Blaga from Ohio State University and his colleagues have executed a technique to take rapid snapshots of molecules in motion, published last week (March 7) in Nature.
By shooting a laser through a tiny hole into a vacuum chamber, Blaga created an infrared laser field. The laser’s intensity knocks an electron out of atomic orbit just for a few femtoseconds, or 10-15 seconds. In that short time before the electron is pulled back into orbit, the bonds holding the molecule’s atoms stretch. And, even if they shift by only a mere angstrom ...












