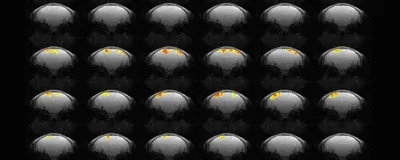
A new approach to magnetic resonance imaging could allow neuroscientists to noninvasively track the propagation of brain signals on millisecond timescales, according to a study published yesterday (October 13) in Science.
The technique, which its creators call “direct imaging of neuronal activity” (DIANA), uses existing magnetic resonance imaging (MRI) technology to take series of quickfire, partial images, and then combines those images to create a high-resolution picture of which bits of the brain are active when.
DIANA has so far only been tested in anesthetized mice, and the mechanisms underlying it aren’t entirely clear, notes Matthew Self, a neuroscientist at the Netherlands Institute for Neuroscience who wasn’t involved in the work. But provided it can be replicated in other labs, the method could represent a “major advance” in brain imaging, he says.
“This would be the first technique which would be able to noninvasively measure neural activity with both a ...