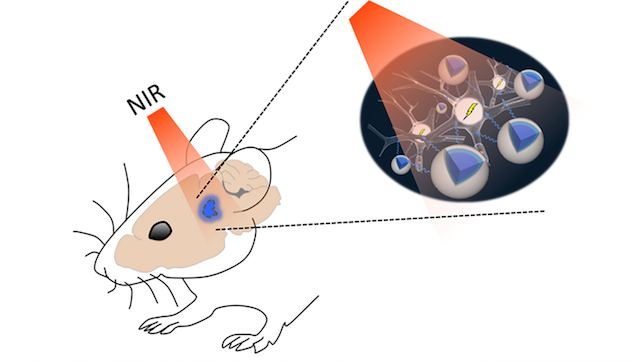
 Schematic showing how near-infrared light can pass through brain tissue and activate neurons via nanoparticles.DR SHUO CHEN AND DR THOMAS J. MCHUGHWhen optogenetics debuted over a decade ago, it quickly became the method of choice for many neuroscientists. By using light to selectively control ion channels on neurons in living animal brains, researchers could see how manipulating specific neural circuits altered behavior in real time. Since then, scientists have used the technique to study brain circuity and function across a variety of species, from fruit flies to monkeys—the method is even being tested in a clinical trial to restore vision in patients with a rare genetic disorder. Today (February 8) in Science, researchers report successfully conducting optogenetics experiments using injected nanoparticles in mice, inching the field closer to a noninvasive method of stimulating the brain with light that could one day have therapeutic uses.
Schematic showing how near-infrared light can pass through brain tissue and activate neurons via nanoparticles.DR SHUO CHEN AND DR THOMAS J. MCHUGHWhen optogenetics debuted over a decade ago, it quickly became the method of choice for many neuroscientists. By using light to selectively control ion channels on neurons in living animal brains, researchers could see how manipulating specific neural circuits altered behavior in real time. Since then, scientists have used the technique to study brain circuity and function across a variety of species, from fruit flies to monkeys—the method is even being tested in a clinical trial to restore vision in patients with a rare genetic disorder. Today (February 8) in Science, researchers report successfully conducting optogenetics experiments using injected nanoparticles in mice, inching the field closer to a noninvasive method of stimulating the brain with light that could one day have therapeutic uses.
“Optogenetics revolutionized how we all do experimental neuroscience in terms of exploring circuits,” says Thomas McHugh, a neuroscientist at the RIKEN Brain Science Institute in Japan. However, this technique currently requires a permanently implanted fiber—so over the last few years, researchers have started to develop ways to stimulate the brain in less invasive ways. A number of groups devised such techniques using magnetic fields, electric currents, and sound.
McHugh and his colleagues decided to try another approach: They chose near-infrared light, which can more easily penetrate tissue than the blue-green light typically used for optogenetics. “What we saw as an advantage was a kind of chemistry-based approach in which we can harness the power of near-infrared light to penetrate tissue, but still use this existing ...






















